Protein drugs were previously made from proteins refined from living organisms. However, they are recently produced using recombinant technology. Insulin, interferon, and erythropoietin are on the market as protein drugs made by recombination.
Confirmation of the expression of recombinant proteins is important from the quality control viewpoint. Methods applied for this include amino acid sequence analysis by peptide sequencer and other simpler methods such as peptide mapping by HPLC or mass mapping by MALDI-TOF MS.
Protein analysis and peptide mass mapping of a model sample of horse heart myoglobin by LC/MS using a quadrupole mass spectrometer is introduced below.
Fig. 1 shows the total ion chromatogram of the trypsin digest of horse heart myoglobin.
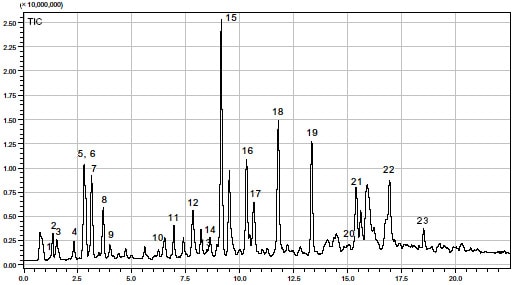
Fig. 1 Total Ion Chromatogram of Trypsin Digest of Myoglobin
Fig. 2 shows the amino acid sequences of peptides derived from horse heart myoglobin obtained using this method. All myoglobin amino acid sequence regions are covered, excepting free lysine. (The recovered peptides are underlined in the diagram.)

Fig. 2 Amino Acid Sequences of Horse Heart Myoglobin
LC/MS is seen to be effective to confirm the expression of recombinant proteins, confirm structural changes in peptides of known structure, and to obtain molecular mass information to supplement the data from a peptide sequencer.
Amino acid composition analysis used in parallel is effective to confirm the composition of peptides containing amino acids. In addition, MS/MS analysis by LCMS-IT-TOF hybrid mass spectrometer is effective for the de novo sequencing of peptides of unknown structure.


